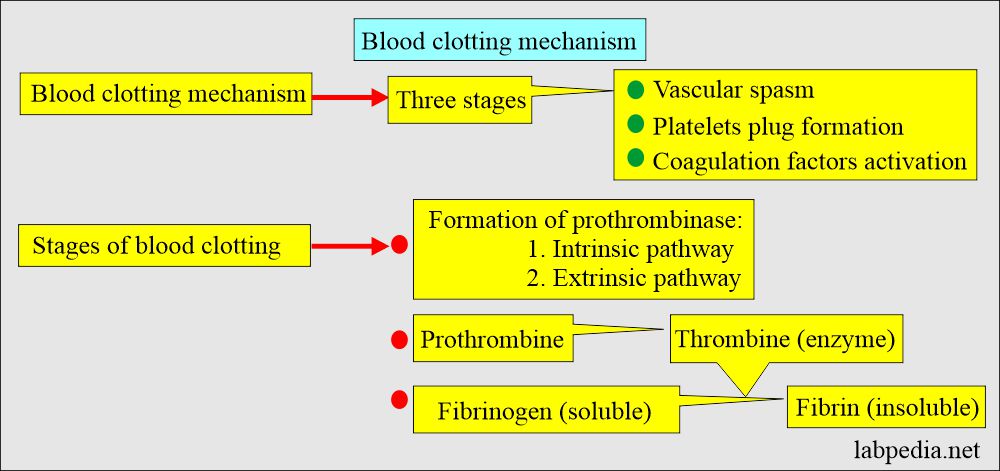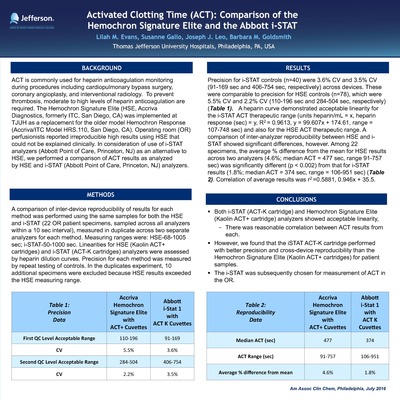
Activated Clotting Time (ACT): Comparison of the Hemochron Signature E" by Lilah Evans, Susanne Gallo et al.

Activated clotting time test alone is inadequate to optimize therapeutic heparin dosage adjustment during post-cardiopulmonary resuscitational extracorporeal membrane oxygenation (e-CPR) | Semantic Scholar
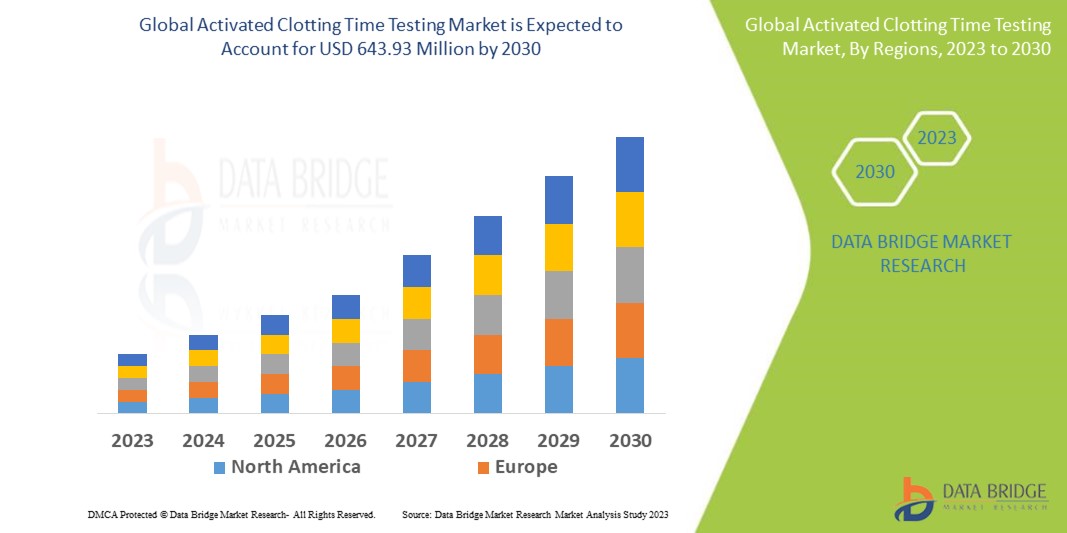
Activated Clotting Time Testing Market – Global Industry Trends and Forecast to 2028 | Data Bridge Market Research

Point-of-care testing of activated clotting time in the ICU: is it relevant? | British Journal of Nursing

Activated Clotting Time (ACT) and Outcomes During PCI for NSTEMI: Insights from FUTURA OASIS-8 | tctmd.com

JCM | Free Full-Text | Running after Activated Clotting Time Values in Patients Receiving Direct Oral Anticoagulants: A Potentially Dangerous Race. Results from a Prospective Study in Atrial Fibrillation Catheter Ablation Procedures

Activated Clotting Time (ACT) and Outcomes During PCI for NSTEMI: Insights from FUTURA OASIS-8 | tctmd.com

Activated Clotting Time Testing Market - Global Industry Analysis, Size, Share, Growth, Trends, and Forecast, 2022 - 2028 - MarketWatch