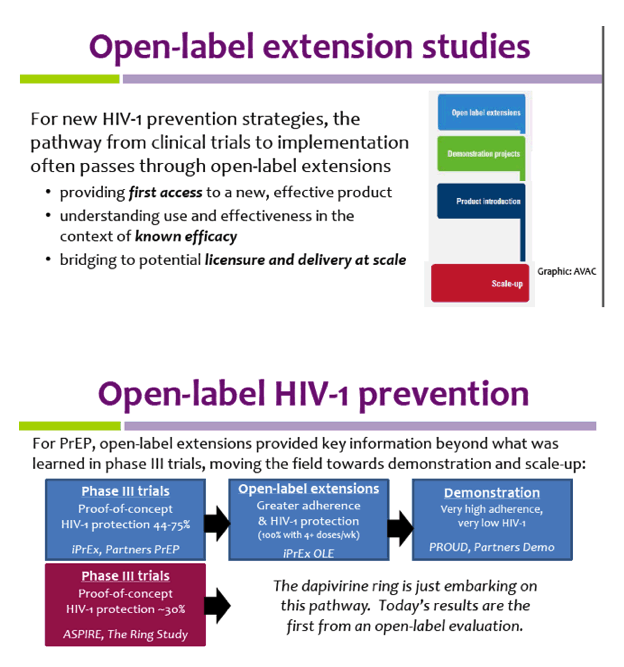
How to process data from clinical trials and their open label extensions PhUSE, Berlin, October 2010 Thomas Grupe and Stephanie Bartsch, Clinical Data. - ppt download

Remdesivir plus standard of care versus standard of care alone for the treatment of patients admitted to hospital with COVID-19 (DisCoVeRy): a phase 3, randomised, controlled, open-label trial - The Lancet Infectious

Study design schematic for the open-label extension study. AE, adverse... | Download Scientific Diagram
PLOS ONE: A Phase I, Open-Label Trial, Evaluating the Safety and Immunogenicity of Candidate Tuberculosis Vaccines AERAS-402 and MVA85A, Administered by Prime-Boost Regime in BCG-Vaccinated Healthy Adults

Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition - Journal of Clinical Epidemiology

Understanding Clinical Trial Terminology: What is an Open Label Clinical Trial? - Concert Pharmaceuticals
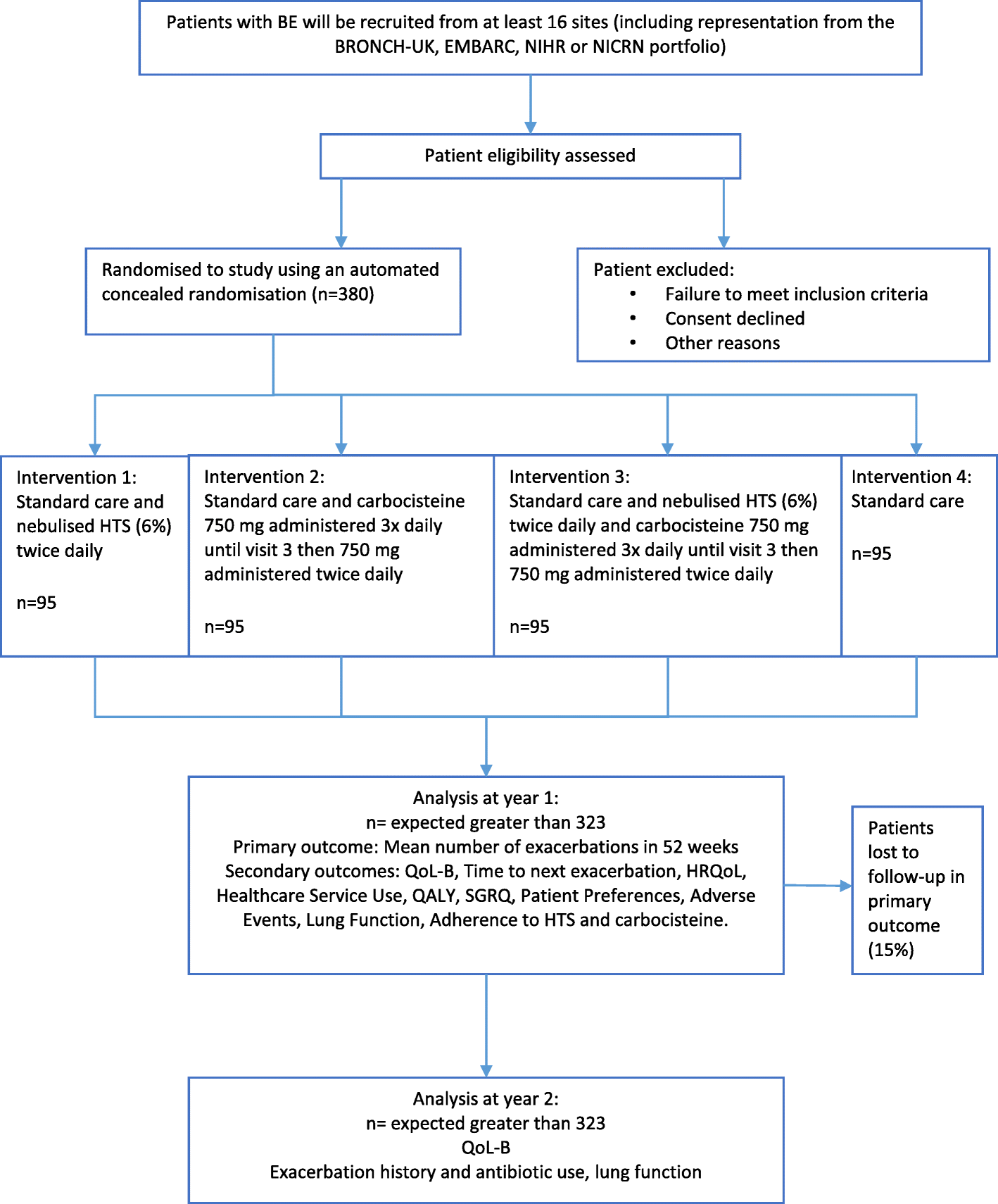
A 2 × 2 factorial, randomised, open-label trial to determine the clinical and cost-effectiveness of hypertonic saline (HTS 6%) and carbocisteine for airway clearance versus usual care over 52 weeks in adults

Effects of antiplatelet therapy on stroke risk by brain imaging features of intracerebral haemorrhage and cerebral small vessel diseases: subgroup analyses of the RESTART randomised, open-label trial - The Lancet Neurology

Rationale and design of a multi-center, open-label, randomised clinical trial comparing HIV incidence and contraceptive benefits in women using three commonly-used contraceptive methods (the ECHO study). | Semantic Scholar

Single center, open label dose escalating trial evaluating once weekly oral ixazomib in ART-suppressed, HIV positive adults and effects on HIV reservoir size in vivo - eClinicalMedicine

Statistical controversies in clinical research: limitations of open-label studies assessing antiangiogenic therapies with regard to evaluation of vascular adverse drug events—a meta-analysis - Annals of Oncology

Phase II single arm open label multicentre clinical trial to evaluate the efficacy and side effects of a combination of gefitinib and methotrexate to treat tubal ectopic pregnancies (GEM II): study protocol
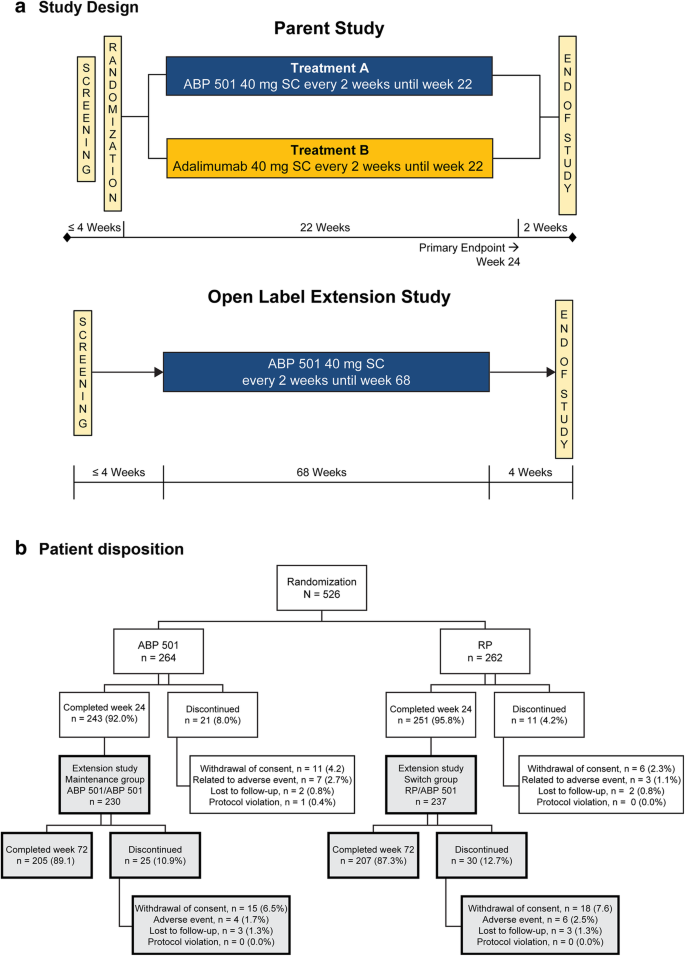
An open-label extension study to demonstrate long-term safety and efficacy of ABP 501 in patients with rheumatoid arthritis | Arthritis Research & Therapy | Full Text
![PDF] Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. | Semantic Scholar PDF] Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/814ad6ccbfa9defca4d2b00c4672f9070cf6b8da/16-Figure1-1.png)
PDF] Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. | Semantic Scholar
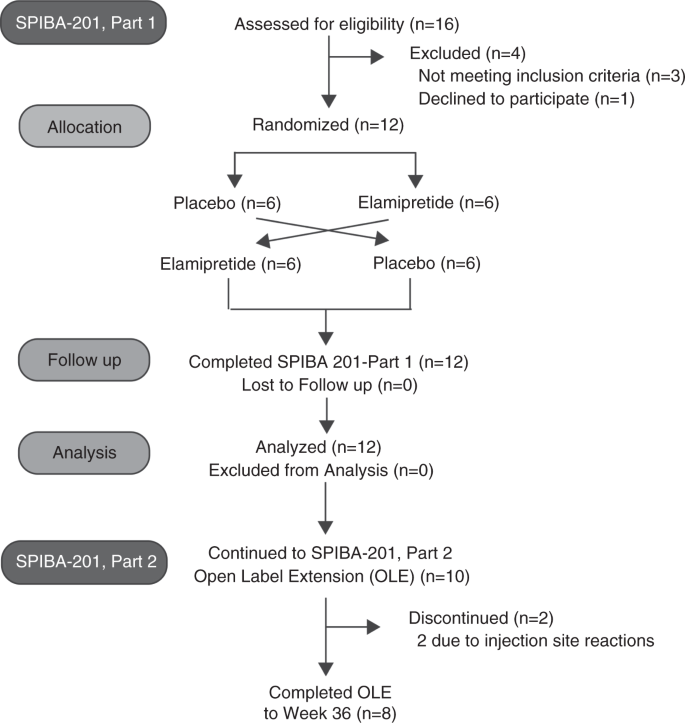
A phase 2/3 randomized clinical trial followed by an open-label extension to evaluate the effectiveness of elamipretide in Barth syndrome, a genetic disorder of mitochondrial cardiolipin metabolism | Genetics in Medicine